Research Interests
Controlling the Lanthanide Coordination Environment
Lanthanides metal compounds offer an intriguing set of physical and chemical properties that differ in many respects from those seen with transition metals. Chemically, the lanthanides are excellent catalysts for a wide range of transformations including, among others: i) addition of N-H and P-H groups to alkenes or alkynes, ii) alkene, lactam and lactone polymerizations, iii) transfer hydrogenation of ketones and imines and iv) Lewis acid catalyzed C-C couplings. Many of these reactions occur at higher rates with different selectivities than for transition metal compounds and the smooth size changes across the lanthanide series allows systematic tuning of compound reactivity. The physical properties of the lanthanides are equally unique and technologically important: i) paramagnetic lanthanide ions are used as commercial contrast agents in magnetic resonance imaging (MRI), ii) near infrared (750-1400 nm) luminescent lanthanide compounds are useful in fiber optic signal transmission and medical imaging and iii) lanthanide radioisotopes that decay by gamma ray emission are suitable for either therapeutic (tumour destruction) or diagnostic (gamma imaging) applications.
In the preceeding applications, control of the lanthanide coordination environment is critical in achieving the desired reactivity or taking advantage of their physical properties. In contrast to the transition metals, the large size and ionic nature of the lanthanides make it very difficult to determine structure and reactivity in advance of synthesis. Specific challenges in catalysis include preventing coordination polymer formation by bridging interactions while still allowing small rective substrates to access the metal centre and creating chiral environments so that highly enantioselective catalysis is possible. In medical applications such as MRI, fluorescence and gamma imaging, compound stability and specificity under physiological conditions are additional concerns that depend on ligand design strategies.
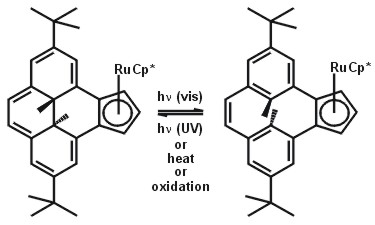
Projects 1 and 2 involve the design of new ligands for organolanthanide chemistry. Designing chiral lanthanide compounds that afford stereochemical control (1) is an important goal; rigid, chiral ligands containing oxazoline functionality are being developed to meet this requirement. Project 2 is aimed at control of the secondary coordination sphere of the lanthanide. Embedding the metal center in a pocket created by large, rigid aromatic ligands is one strategy to prevent bridging interactions (that often severely limit reactivity) without limiting small substrate access. Project 3, with Professor Reg Mitchell, is exploring the effect of metal coordination on the photoswitching properties of the dimethyldihydropyrene (DHP) – cyclophanediene (CPD) photoswitch developed by the Mitchell group. This project is not focused on lanthanide chemistry but it is related to Project 2 in that it explores the use of very large aromatic systems as ligands towards metals. There is also an ongoing effort in developing lanthanide complexes suitable for gamma and luminescence imaging medical applications - this project is confidential at this stage. The ‘designer ligand’ strategy involved in our research program requires extensive organic and inorganic synthesis including the handling of extremely air and moisture sensitive species. Prospective graduate students with good synthetic skills are needed for all projects - contact me for details!
 |
 |
|||
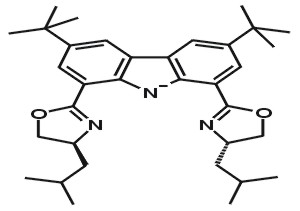
| Chiral carbazole bis(oxazoline) ligands | ||||
R,R-i-Bu2Carbox |
||||
 |
 |
|||
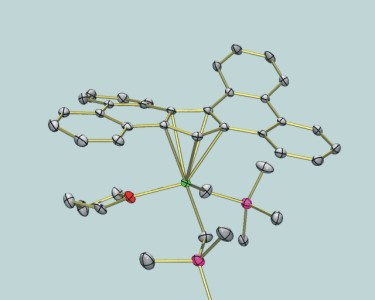
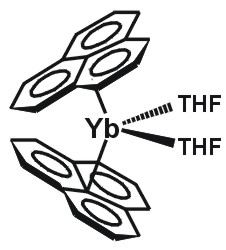
| Large aromatic-fused ligands | ||||
YCp'(CH2SiMe3)2(THF) |
(Phenalenide)2Yb(THF)2 |
|||
 |
 |
|||
| Photoswitchable DHP-metal complexes (with R.H. Mitchell) | ||||
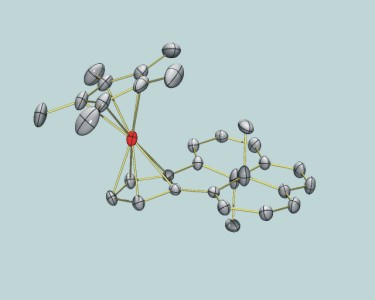
RuCp*(DHPCp) |
||||